 Health & Physiology
Health & Physiology
A special delivery for malaria-transmitting mosquitoes
Female mosquitoes pass on malaria parasites that infect hundreds of millions of people each year. Despite the progress we have made to prevent this disease, chemical insecticides are not as effective as they used to be. To fight against this life-threatening disease, we have developed a fungus that effectively kills wild-caught mosquitoes and hampers their reproduction.
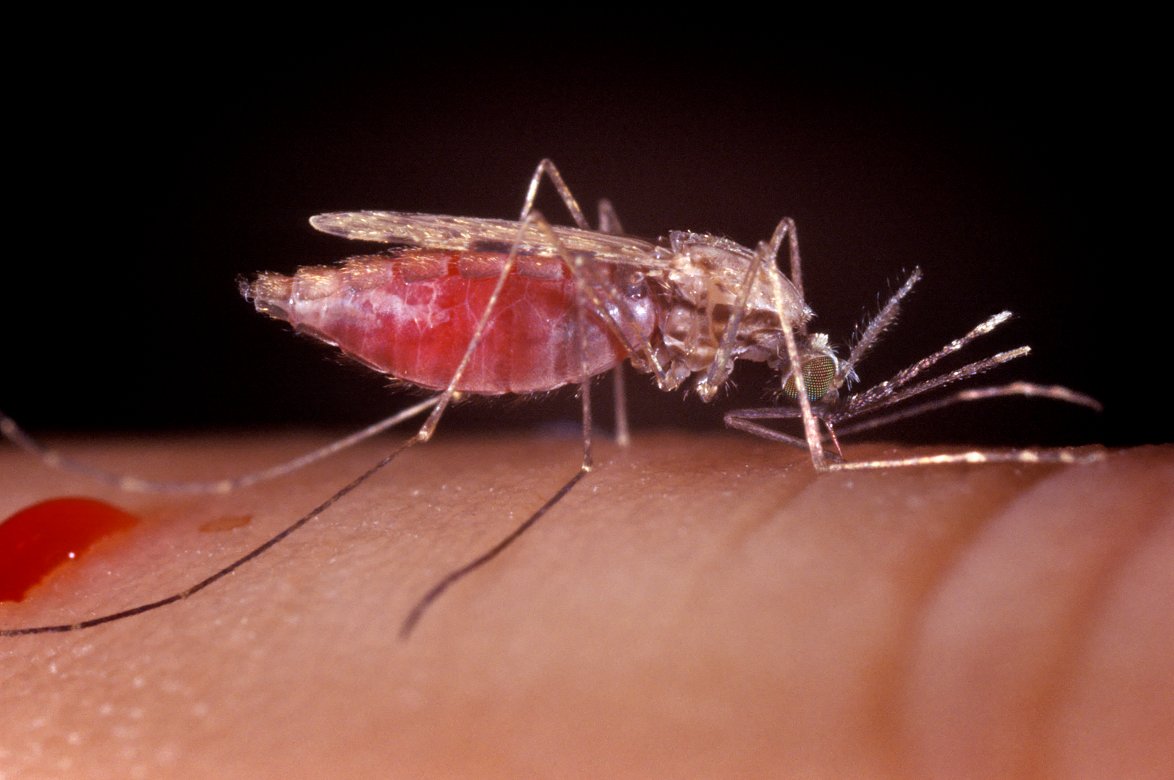
Infected female Anopheles mosquitoes deliver malaria parasites to hundreds of millions of people worldwide, killing nearly half a million people every year. In the past two decades, we have successfully decreased the global malaria burden by half thanks to chemical insecticides, which are sprayed in houses and applied to bed nets. Unfortunately, we did not have much time to savor this victory, as our progress has stalled in recent years.
The efforts have stalled because malaria mosquitoes became resistant to chemical insecticides. Imagine that these mosquitoes are a taxi, delivering their passengers, malaria parasites, to a new person. In 2000, we used chemical pesticides as an effective roadblock, stopping these taxis in their tracks. Today, many mosquitoes can simply avoid this roadblock: this means that chemical insecticides, our primary tool to control mosquitoes, are not able to hold back these malaria carriers like they used to. We need more tools to stop mosquitoes from delivering malaria parasites to new people.
We are developing a new tool that improves on a natural fungal pathogen of mosquitoes. This pathogen, called Metarhizium pingshaense, has a taste for mosquitoes, just like some mosquitoes have a taste for us. We already use close relatives to this fungus worldwide to control insects that eat our crops. They are common in nature and are safe for us.
If Metarhizium tastes a mosquito, it will burrow into its blood. However, this is only half of the battle, as the mosquito's blood is an unsafe place for a fungus. To protect itself from mosquito's immune system, the fungus cloaks itself in a protein that makes it invisible to the mosquito. With its cloak on, the fungus will slowly multiply until it kills the mosquito.
Impressive as they are naturally, the wild fungus does not kill mosquitoes fast enough to control them effectively. If Metarhizium infects a mosquito that will soon be ready to deliver malaria parasites to someone, these parasites may still make their way into the next person; moreover, the female mosquito may even have time to lay eggs. When it comes to human diseases, we do not have time to wait.
Fortunately, there are many ways that we can speed up these fungi to help us control mosquitoes. The most promising option involves specific toxins that we can borrow from spiders. Even though the words "spider toxin" may sound scary, spiders produce a range of proteins in their venom, and some of these proteins are toxic only to insects. In our studies, we have borrowed a toxin called Hybrid produced by the Blue Mountains funnel-web spider native to Australia.
Using the right tools in the lab, we can make the fungus produce the Hybrid toxin only once it has reached mosquito blood and dons its invisibility cloak. Here, we enabled a natural mosquito pathogen to deliver the insect-specific toxin from a spider into mosquito's blood.
In a collaboration between the labs in Burkina Faso and the US, we found that this super-charged fungus is more effective against mosquitoes than the natural fungus. To see if this works in field conditions, we tested this new fungus against wild-caught mosquitoes in a special facility called a MosquitoSphere. This MosquitoSphere has separate compartments for the experiments that mimic natural field conditions.
While testing the efficacy of the Hybrid toxin in the MosquitoSphere, we found that it kills more mosquitoes and kills them faster than the normal fungus. Moreover, this new fungus dampens the reproduction of infected mosquitoes.
We believe that the path forward in our effort to control malaria is the one where scientists, communities, and technologies work together. This project is an exciting development for this emerging fungal biotechnology, and it provides an additional tool for us to stop mosquitoes from carrying malaria from a sick person to a healthy one. Even though we have been struggling against malaria parasites and these mosquitoes that carry them since the dawn of our civilization, new technologies like this one offer a hopeful glimpse into our malaria-free future.
Original Article:
Lovett B, Bilgo E, Millogo S et al. Transgenic Metarhizium rapidly kills mosquitoes in a malaria-endemic region of Burkina Faso. Science. 2019;364(6443):894-897Next read: DNA of Things: how a plastic bunny got DNA by Julian Koch , Robert N. Grass
Edited by:
Dr. Monika Stankova , Senior Scientific Editor
We thought you might like
Driving down malaria
Jul 18, 2017 in Health & Physiology | 4 min read by Andrew Hammond , Xenia Karlsson , Ziyin WangHow lab-grown blood vessels can help us understand malaria
Oct 28, 2020 in Maths, Physics & Chemistry | 3.5 min read by Caitlin Howard , Joseph D. Smith , Ying ZhengThe Trojan mosquito: an in-house parasite defends against malaria
Jan 26, 2021 in Health & Physiology | 3 min read by Enock Mukibetti Mararo , Lilian Mbaisi , Edward Makhulu EdmondThe taste for human sweat
Nov 4, 2019 in Evolution & Behaviour | 3 min read by John S. Castillo , Matthew DeGennaroMore from Health & Physiology
How obesity can improve the efficacy of cancer treatment: role of the sex hormone estrogens.
Dec 3, 2025 in Health & Physiology | 3.5 min read by Eloïse Dupuychaffray , Carole BourquinTobacco smoking and other exposures shut off cancer-fighting genes
Aug 31, 2024 in Health & Physiology | 3 min read by Jüri Reimand , Nina AdlerA hidden clock that times cytoplasmic divisions
Aug 30, 2024 in Health & Physiology | 3 min read by Cindy OwWhen two kinases go for a dance
Aug 2, 2024 in Health & Physiology | 4 min read by Ioannis Galdadas , Francesco Luigi Gervasio , Pauline JuyouxAwakening the thymus to cure SARS-CoV-2 infection: a matter of genes
Jul 27, 2024 in Health & Physiology | 3.5 min read by Stefano Marullo , Cheynier RemiEditor's picks
Trending now
Popular topics


